Better Outcomes.
Simplified.
Carotid
Extra Stroke Protection When It Matters Most
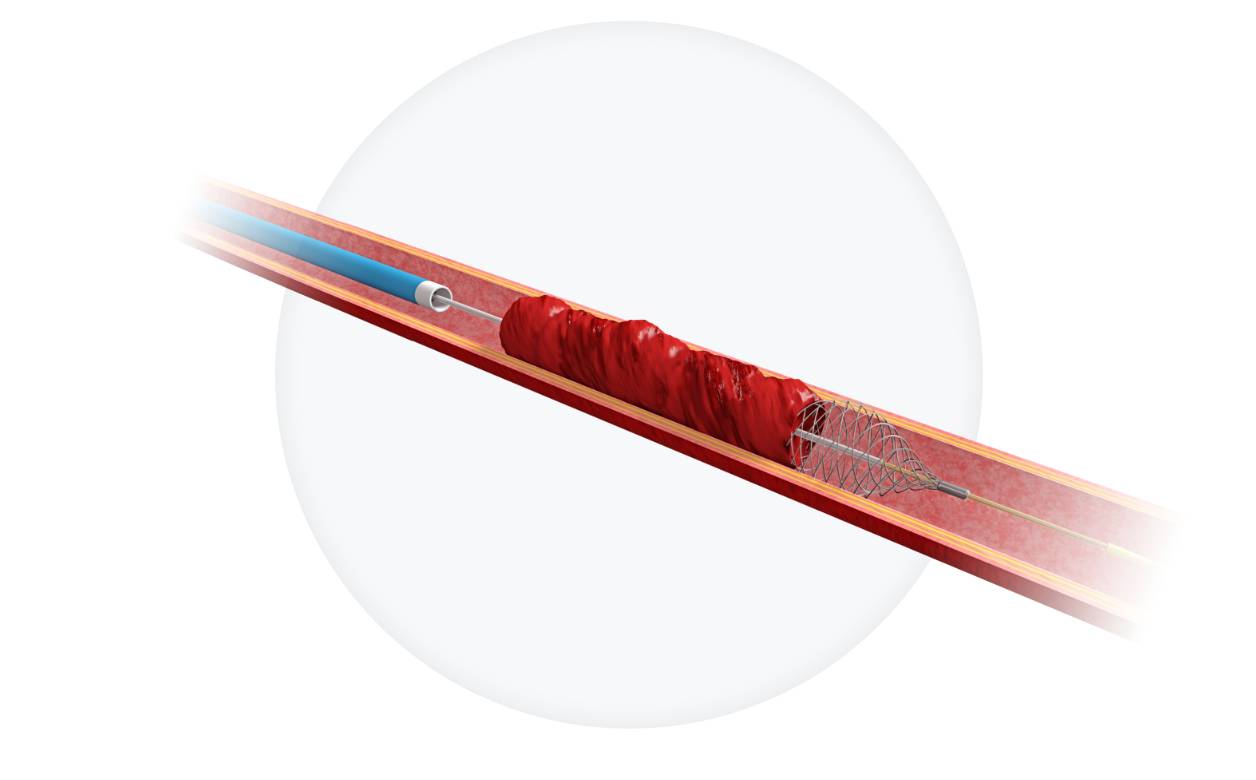
- Designed to safeguard against peri-procedural stroke and long-term cognitive impairment associated with micro-embolization1-3
- Captures more micro-embolic debris than traditional filters4-6
- Streamlined procedure with 2-in-1 technology
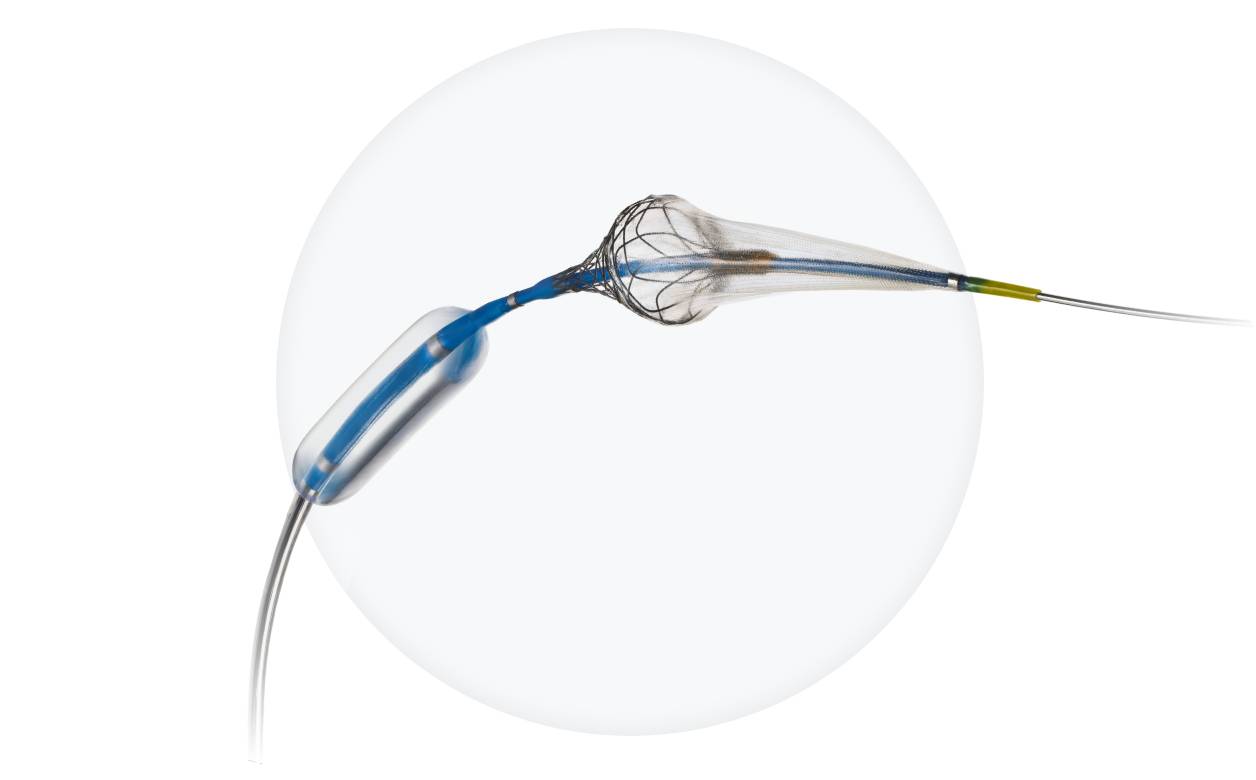
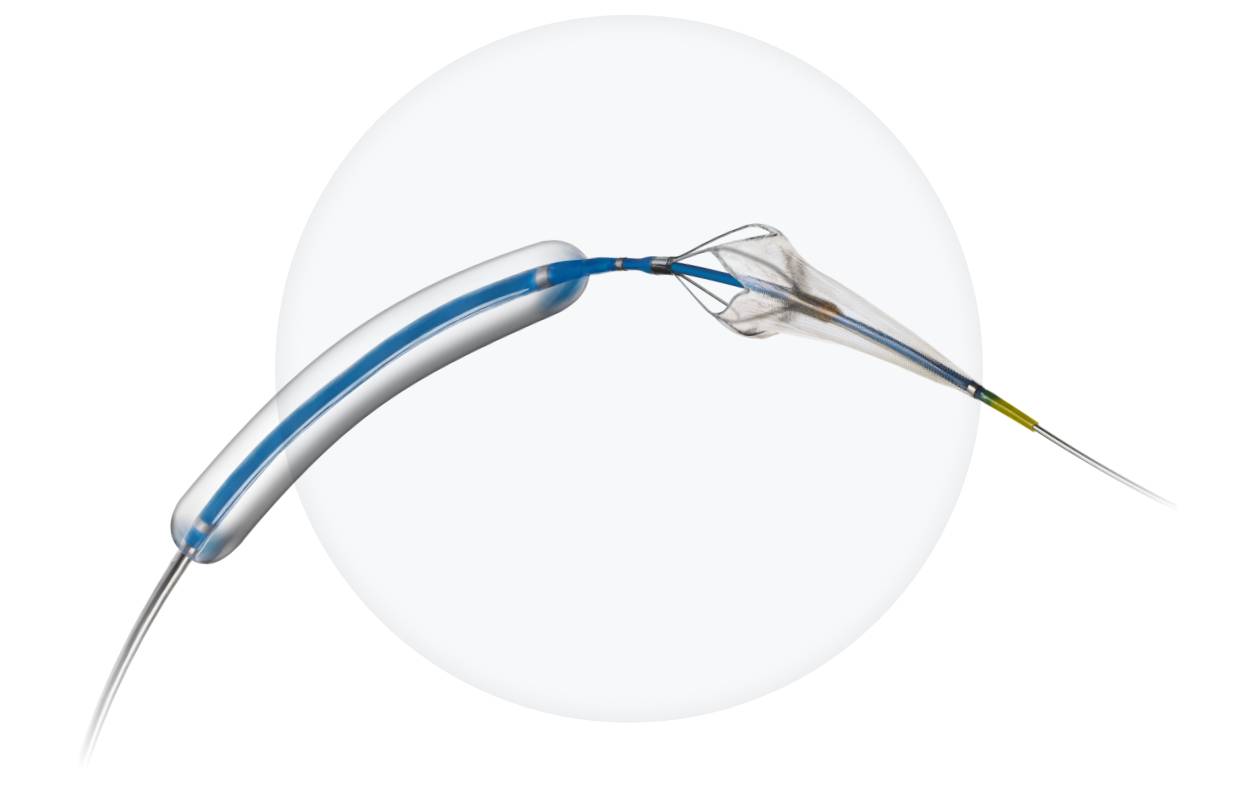
Advanced Stroke Protection
- Leverages Paladin® platform + optimized carotid stent
- Plaque coverage AND flexibility with FlexRing™ technology
- Fewer procedural steps than traditional stents with 3-in-1 technology
CAUTION: Investigational device. Limited by Federal (United States) law to investigational use.
Peripheral Vascular